
use 4.184 j/g°c as the specific heat of water. Barium hydroxide is the precipitate in the reaction. Forming a precipitate when Al2 (SO4)3 is added to NaOH. Best Answer Copy Precipitate forms when an INSOLUBLE substance is formed. As an example, PbCl 2 is a white precipitate and PbI 2 is a yellow precipitate. Is PbCl2 a precipitate? So, consider the three compounds that you have: BaCl2 added to a solution containing Na2CO3 and NaOH.
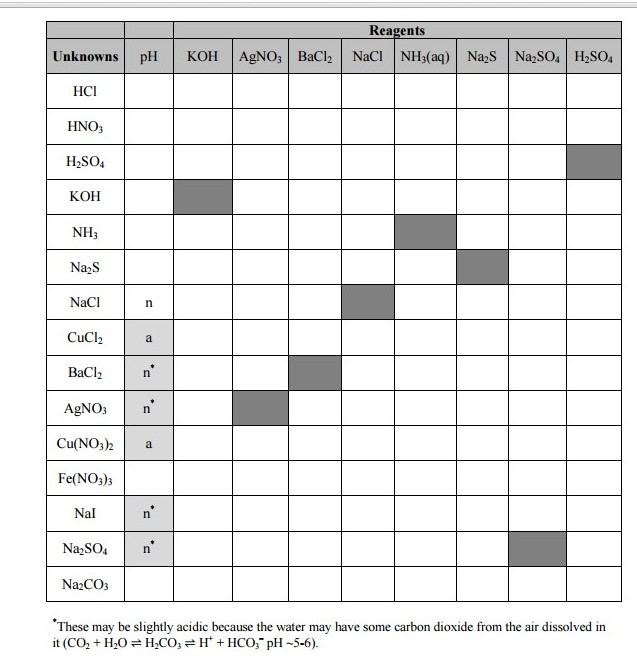
The sodium ions in the solution will not form a precipitate with the chloride ions from barium chloride, as sodium salts (alkali metal salts) are always very soluble in water. 13 What is the balanced equation for AgNO3 NaCl? What is the product of BaCl2 NaOH? Why does copper sulfate and sodium hydroxide form a precipitate? (e). HBr + NaOH Na2S + HF Li2CO3 + HNO3 MgCl2 + KOH All of these solution pairs will produce a precipitate. Use this information to suggest likely ionic formulas for the precipitates observed. Will not forma precipitate K2SO4+BaCl2 NaOH + Mgl2 Table Question: Question 2 (2 points) Determine if the reactants shown on the right would result in the formation of a precipitate. OK processing pottery grade barium carbonate by first converting to the chloride. 11 Does NaCl and AgNO3 form a precipitate? 14 How do you balance chemical equations? Precipitations Reactions. Over time, sodium hydroxide Barium chloride reacts with sodium phosphate to form barium phosphate and sodium chloride. There are many common types of reactions that occur in aqueous solutions. In some steps such as steps a, b, and d in the BaCl2 solution, the precipitate formed at the bottom of the test tube when the precipitate was re-dissolved. Considering that CaCl2 and Na2SO4 is water soluble, and CaSO4 is mostly insoluble in wat.


Simple equation of barium chloride and copper (II) sulfate If white precipitate forms then continue to add NaOH to it and observe whether the precipitate dissolves. This is a precipitation reaction: BaSO 4 is the formed precipitate. There are three main steps for writing the net ionic equation for NaNO3 + CaCl2 = NaCl + Ca(NO3)2 (Sodium nitrate + Calcium chloride). Barium stimulates striated, cardiac, and smooth muscle, regardless of. The reaction takes place in aqueous solution. Barium chloride is salt that dissolves in water, sodium hydroxide is strong base that dissolves in water. Your reasoning is quite right 208.22 g BaCl2 yields 233.39g BASO4 1g BaCl2 yields (233.39/208.22)g BaSO4 Therefore, the precipitate is the insoluble solid that forms during the chemical reaction and separates out in solution. 18 Does AgNO3 and Ki form a precipitate? The precipitation reaction is one of them, which results in the formation of precipitate (insoluble product). This type of reaction is called a precipitation reaction, and the solid produced in the reaction is known as the precipitate. Which combination will result in a precipitate forming? Six Bottles: an Experiment in Chemical Identification. Addition of NaOH, Na2O2 to Cr3+ solution forms Na2CrO4, a yellow solution. Therefore, the precipitate is the insoluble solid that forms during the chemical reaction and separates out in solution. What is the color of precipitate formed by CaCl 2 + NaOH. BaCl2 reacts with KOH forming KCl which is a salt and Ba(OH)2 which is the precipitate. HCl(aq) + NaOH(aq) H 2O(l) + NaCl(aq) In this reaction, hydrochloric acid joins with sodium hydroxide, a base. BaCl2 + 2NaOH → Ba (OH)2 + 2NaCl The reaction of barium chloride with sodium hydroxide with the result of hydroxide precipitate barium. When a combination of ions is described as insoluble, a precipitate forms. That insoluble salt is known as precipitate and therefore such reactions are named precipitation reactions.An example of precipitation reaction is, Precipitates have different colours.
